
5.16
US FDA approval of Innolife's first new drug INL1 entering the phase II clinical trial
The first new drug, which was developed by Innolife to treat heart failure and associated indications, has been approved for the direct access to phase II clinical trial by US FDA. INL1 achieved regenerative effect based on its novel mechanism of acting as a carrier to replenish copper specifically to ischemic injured heart tissue, thereby, launching the inherent tissue injury repair system of human body. INL1 will be a great breakthrough in the heart failure therapy.

2.24
University of Tennessee Health Science Center expert delegation visiting Innolife
Recently, Dr. Steven R. Goodman, the vice chancellor of the University of Tennessee Health Sciences Center, led an expert delegation to visit Innolife and discuss the potential cooperation opportunities. During the meeting, the Tennessee experts sensed Innolife’s actual strength of new drug research and development, product lines, and the progress of our first new drug INL1 which was applied for Phase II clinical trials to FDA in US, meanwhile, they also introduced the clinical resource advantages of the University of Tennessee Health Sciences Center. After the communication and discussion, a preliminary cooperative agreement about INL1 project has been achieved between the two sides.
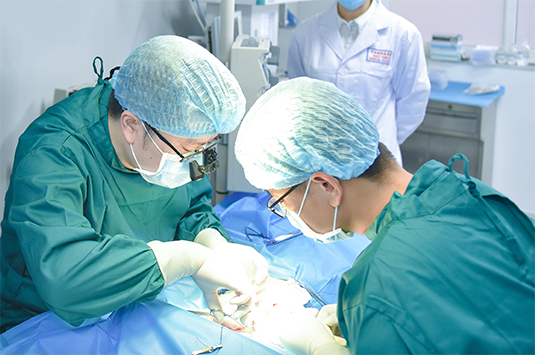
12.11
Dr. Kang's 3D bioprinting team have successfully implanted 3D bioprinted blood vessels made from stem cells into monkeys
Dr. Kang's 3D bioprinting team have successfully implanted 3D bioprinted blood vessels made from stem cells into rhesus monkeys, realized the regeneration of blood vessel, marking an important step towards printing blood vessels and other organs for human transplants. "It is groundbreaking work that will change the way regenerative medicine will develop," said Sir Alfred Cuschieri, a professor at the University of Dundee. This achievement could benefit the 156 million people every year worldwide who need artificial blood vessels or vessel support structures.

9.30
Innolife admission to Jingrong Center, Tianfu New Area
Innolife signed the agreement with the Management Committee of Tianfu New Area, becoming one of the enterprises firstly moving into Science City nearby Xinglong lake, and attended the opening ceremony Jingrong center.

6.30
Pre-IND meeting with the U.S. FDA
Innolife research team representatives attended Pre-IND meeting with the U.S. FDA for the IND of INL1 project with new indication of ischemic heart failure. The Division made several favorable comments about INL1 and mentioned that INL1 was eligible for Fast Track designation. If the result of Phase II is positive, INL1 would be a major breakthrough in the field of treatment of ischemic heart disease.

8.18
Establishment of Innolife Co., Ltd.
Innolife is one of enterprises firstly set up in Tianfu New Area, Chengdu. The Company is dedicated to provide innovations and solutions for regeneration of failure organs by discovering, developing original products to mobilize the human body inherent self-repair capability to finally realize the rejuvenation and solve the most challenging medical problems.

 English
English 简体中文
简体中文



